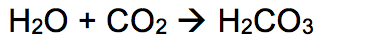Discovery of Carbonic Acid
Carbonic acid was first discovered by Dr. Brownrigg around the year 1757. The gas form of carbonic acid, however, was discovered by Joseph Black; a Scottish chemist and physician.
It was found in the Pyrmont Water, Austrailia. The name Carbonic Acid was given to the molecule in 1787, roughly 30 years later.
It was found in the Pyrmont Water, Austrailia. The name Carbonic Acid was given to the molecule in 1787, roughly 30 years later.
Found in Nature
Artificially Made and Chemical Creation
|
Carbonic Acid is also used to fabricate these products/uses:
In sodas and other carbonated drinks, dissolved carbon dioxide (CO2) is added to water (H2O). This creates the Carbonic Acid. In addition to this, Phosphoric Acid is also added to the soda as a 'buffer' to keep the pH level at a more tolerable level. In general, water and carbon dioxide are reactants to create carbonic acid. This equation can be seen to the right. |
Exposure and Effects
|
Carbonic is safe for human consumption and exposure when it is within a tolerable acidity. Soda has an acidity with a pH level of around 3. As mentioned in the section before, Phosphoric Acid is added to raise the pH level of the soda (thus lowering it's acidity).
Carbonic Acid, however, does corrode matter over time. Caves under the surface of the earth can be carved out by Carbonic Acid over a span of many years. In addition to this, Carbonic Acid can also corrode things in the form of acid rain. The picture to the right shows how over time, acid rain can deteriorate structures like the statue. |