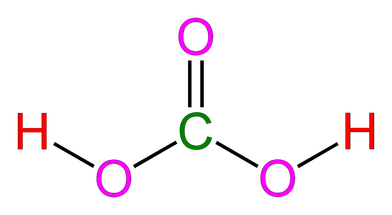
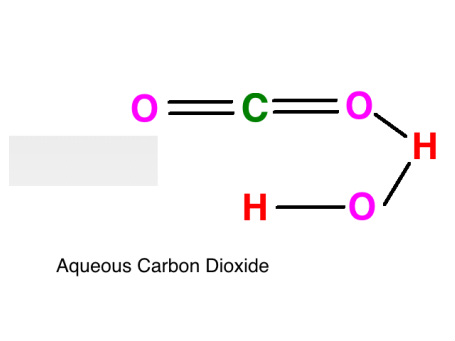
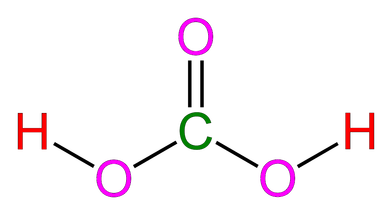
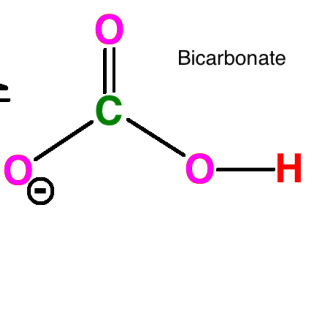
Carbonic Acid's Interaction with Aqueous Carbon Dioxide
Both carbonic acid and aqueous carbon dioxide are polar. The hydrogen is attracted to the oxygen, thus creating a hydrogen bond between the two molecules. The H+ are attracted to the O- In addition to this, dispersion forces exist between any two molecules in a liquid.
Carbonic Acid's Interaction with Bicarbonate
Both the carbonic acid molecule and the bicarbonate molecule are polar. Hydrogen bonds occur because the positive hydrogen (H) atoms are attracted to the negative oxygen (O) in the opposite molecule. This forms a hydrogen bond between the oxygen and hydrogen. In addition to this, dispersion forces exist between any two molecules in a liquid.
Key:
Red = Hydrogen (H)
Pink = Oxygen (O)
Green = Carbon (C)
Pink = Oxygen (O)
Green = Carbon (C)